随着人类对癌症研究的不断深入,发现肿瘤的预后与其分期、分级、恶性程度等因素密切相关。肿瘤免疫微环境(TME)与肿瘤的发生发展、复发、转移和耐药等进展密切相关。研究表明,TME中肿瘤浸润淋巴细胞(TILs)与肿瘤细胞作用密切,也是肿瘤预后的有利因素,TILs的浸润程度可反映肿瘤患者的预后及疾病进程[1]。
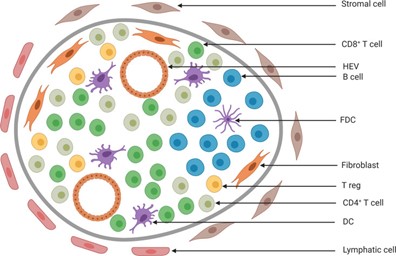
图1 TLS的组成模型
三级淋巴结构(TLS)是肿瘤中慢性炎症刺激部位TILs的聚集体,主要由B细胞、T细胞和树突状细胞组成,并显示出不同层次的组织,从局部集中的免疫细胞聚集体,到明确定义的B细胞滤泡,再到包含生发中心的成熟滤泡。TLS的浸润与实体瘤患者癌症免疫疗法的生存率和临床结果的改善密切相关,是潜在的预后性分子标志物[2]。三级淋巴结构的细胞组成、所处位置、密度、成熟状态等相关因素被证明对肺癌、肠癌、乳腺癌、肝癌、胃癌、头颈癌、卵巢癌、黑色素瘤等多种肿瘤的预后具有评估作用。研究表明,对大多数肿瘤而言,TME中TLS的浸润对肿瘤的预后有利,TLS密度越高、浸润程度越高、成熟度越高,患者预后越好。但肿瘤存在异质性,不同类型肿瘤的免疫细胞的浸润程度也存在差异,TLS的预后价值也不尽相同[3]。
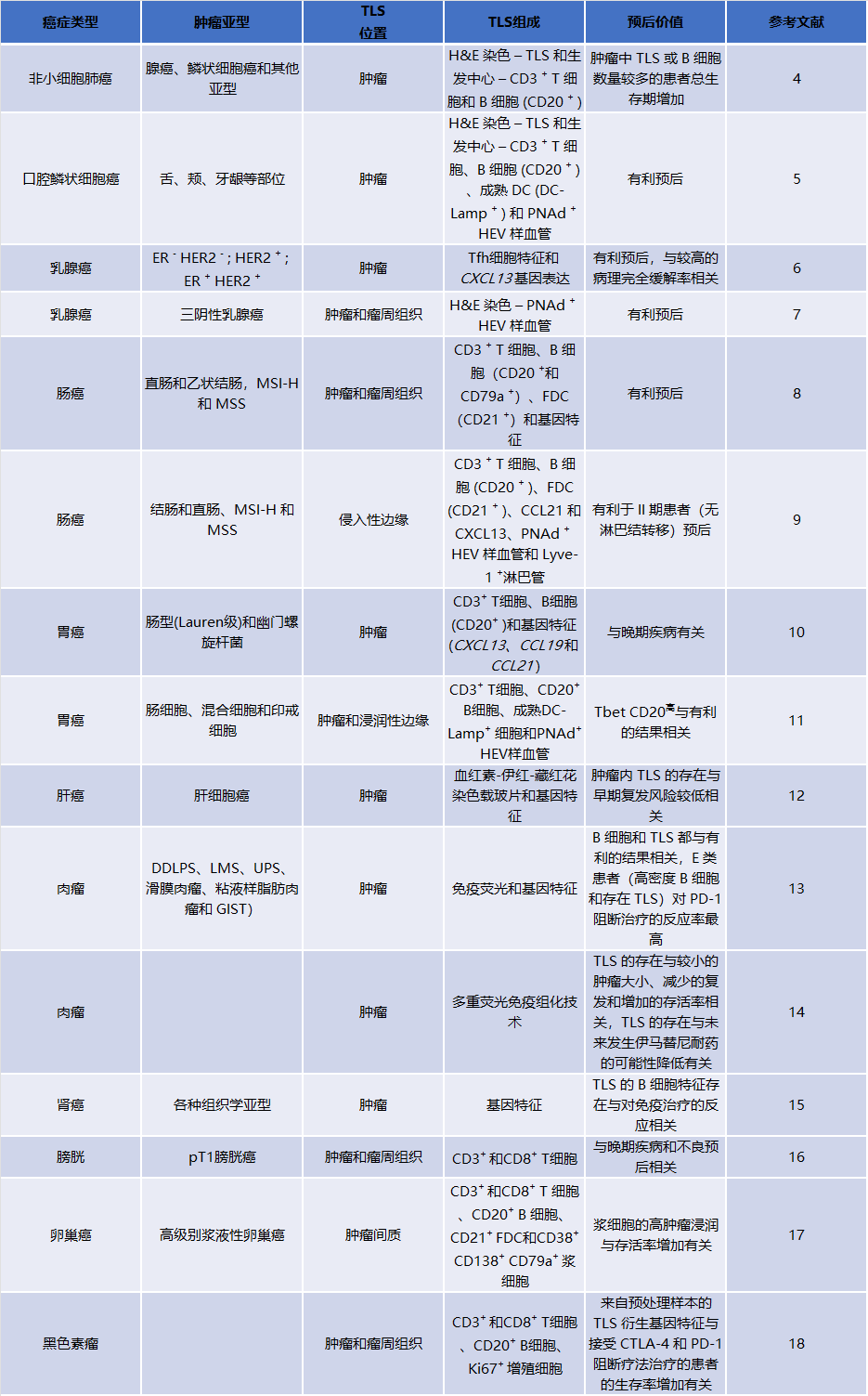
表1:TLS在不同类型肿瘤中的预后价值
随着免疫治疗陆续进入肺癌、肠癌、胃癌等多癌种的一线治疗方案,寻求精准有效的预测免疫治疗疗效的生物标志物成为临床上亟需解决的问题。《自然》(Nature)期刊上曾连发三篇关于三级淋巴结构(TLS)在黑色素瘤、肾癌和肉瘤中的免疫治疗领域的研究,三项研究共同认为:TLS与免疫治疗的预后密切相关,甚至患者能否能从抗PD-1免疫治疗中获益,可能也取决于TLS的浸润情况[13, 15, 18]。此后,评估TLS预测免疫治疗疗效价值的研究相继开展。近期在《自然·癌症》(Nature Cancer)期刊上发表的研究(发现队列n=328)探索了TLS状态与免疫治疗应答率、无进展生存(PFS)和总生存(OS)的关系,探究PD-L1表达水平、CD8+T细胞密度与TLS状态的关系。采用多重免疫组化技术(mIHC)结合CD8、CD20、CD21和CD23等标记物判断TLS成熟度。研究表明CD8+T细胞高浸润与TLS可独立于PD-L1表达预测免疫检查点抑制剂的疗效,甚至可以提高PD-L1阴性患者获益的可能性[19]。
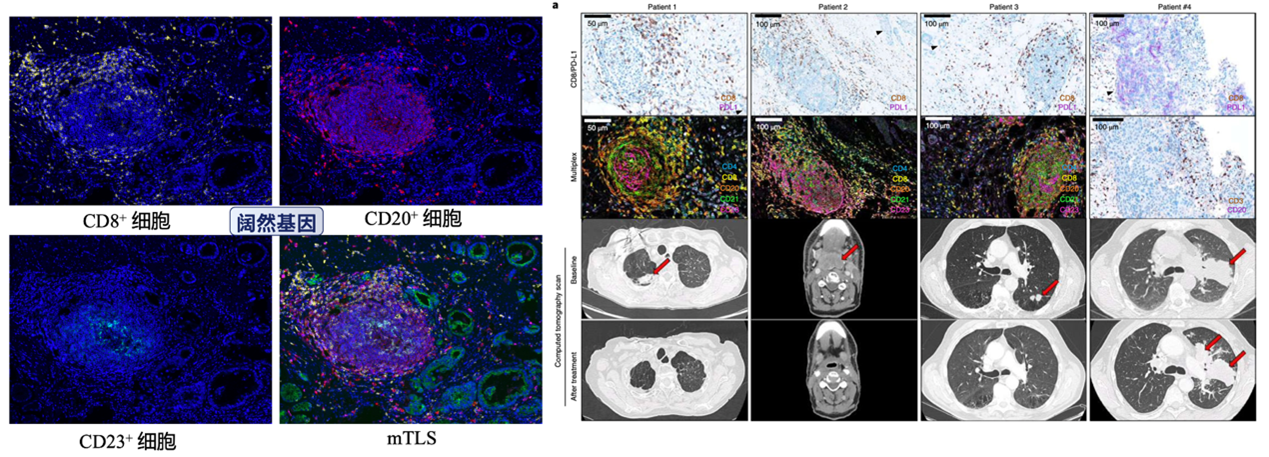
图2 TLS可独立于PD-L1表达预测免疫检查点抑制剂的疗效
研究发现,与TLS相关的细胞类型较多,且结构复杂。如何从组织中识别检测和量化TLS是目前的热点问题[20]。目前的识别方式主要有:
①通过HE染色后显微镜下识别致密淋巴组织聚集物;
②通过免疫组化对TLS的免疫细胞选择性地显示和定量;
③通过免疫荧光标记TLS相关细胞进行识别和定量分析;
④基因表达谱检测TLS相关趋化因子。
其中,多重荧光免疫组化技术(mIHC)可以在一张组织切片上标记多种TLS相关的免疫细胞,直观地表征TLS结构。通过对不同肿瘤免疫细胞的标记,可以区分不同亚型和成熟度的TLS,结合数据分析软件,还可以定量评估TLS的密度、位置等信息。因此,mIHC是目前TLS研究领域中最为常用的技术之一[13,15,18],有助于深入研究肿瘤免疫微环境,为后续精准免疫治疗提供更多临床证据。
综上所述,TLS作为免疫系统的组成部分,可发挥有效的抗肿瘤作用,促进T细胞传递至不可达的肿瘤区域[2]。TLS的存在是肿瘤患者良好预后的指标,是潜在的下一代预测标志物和治疗靶点。临床上广泛开展TLS的研究,有助于制定有效的免疫治疗策略并推动新药的研发[3,20]。
1.Anderson NM, Simon MC. The tumor microenvironment. Curr Biol. 2020 Aug 17;30(16):R921-R925. doi: 10.1016/j.cub.2020.06.081. PMID: 32810447; PMCID: PMC8194051.
2.Catherine S, Florent P, et al. Tertiary lymphoid structures in the era of cancer immunotherapy. Nature Reviews Cancer, 2019, 19: 307–325.
3.N J, J T, Sl N, Gt B. Tertiary lymphoid structures and B lymphocytes in cancer prognosis and response to immunotherapies. Oncoimmunology. 2021 Mar 29;10(1):1900508. doi: 10.1080/2162402X.2021.1900508. PMID: 33854820; PMCID: PMC8018489.
4.Tang J. B Cells and tertiary lymphoid structures influence survival in lung cancer patients with resectable tumors. Cancers (Basel). 2020;12(9). doi:10.3390/cancers12092644.
5.Li Q. Prognostic value of tertiary lymphoid structure and tumour infiltrating lymphocytes in oral squamous cell carcinoma. Int J Oral Sci. 2020;12(1):24.
6.Gu-Trantien C. CD4(+) follicular helper T cell infiltration predicts breast cancer survival. J Clin Invest. 2013;123(7):2873–2892. doi:10.1172/JCI67428.
7.Lee HJ. Tertiary lymphoid structures: prognostic significance and relationship with tumour-infiltrating lymphocytes in triple-negative breast cancer. J Clin Pathol. 2016;69(5):422–430.
8.Coppola D. Unique ectopic lymph node-like structures present in human primary colorectal carcinoma are identified by immune gene array profiling. Am J Pathol. 2011;179(1):37–45.
9.Di Caro G. Occurrence of tertiary lymphoid tissue is associated with T-cell infiltration and predicts better prognosis in early-stage colorectal cancers. Clin Cancer Res. 2014;20(8):2147–2158.
10.Hill DG. Hyperactive gp130/STAT3-driven gastric tumourigenesis promotes submucosal tertiary lymphoid structure development. Int J Cancer. 2018;143(1):167–178.
11.Hennequin A. Tumor infiltration by Tbet+ effector T cells and CD20+ B cells is associated with survival in gastric cancer patients. Oncoimmunology. 2016;5(2):e1054598.
12.Calderaro J. Intra-tumoral tertiary lymphoid structures are associated with a low risk of early recurrence of hepatocellular carcinoma. J Hepatol. 2019;70(1):58–65.
13.Petitprez F. B cells are associated with survival and immunotherapy response in sarcoma. Nature. 2020;577(7791):556–560.
14.Lin Q. Tumor-associated tertiary lymphoid structure predicts post operative outcomes in patients with primary gastrointestinal stromal tumors. Oncoimmunology. 2020;9(1):1747339.
15.Helmink BA. B cells and tertiary lymphoid structures promote immunotherapy response. Nature. 2020;577(7791):549–555.
16.Hulsen S. High stroma T-Cell infiltration is associated with better survival in stage pT1 bladder cancer. Int J Mol Sci. 2020;21(21).
17.Kroeger DR, Milne K, Nelson BH. Tumor-infiltrating plasma cells are associated with tertiary lymphoid structures, cytolytic T-Cell responses, and superior prognosis in ovarian cancer. Clin Cancer Res. 2016;22(12):3005–3015.
18.Cabrita R. Tertiary lymphoid structures improve immunotherapy and survival in melanoma. Nature. 2020;577(7791):561–565.
19.Vanhersecke, L. et al. Mature tertiary lymphoid structures predict immune checkpoint inhibitor efficacy in solid tumors independently of PD-L1 expression. Nature Caner, 2021, 2, 794–802.
20.Schumacher TN, Thommen DS. Tertiary lymphoid structures in cancer. Science. 2022 Jan 7;375(6576):eabf9419. doi: 10.1126/science.abf9419. Epub 2022 Jan 7. PMID: 34990248.

排版编辑:Halo













 苏公网安备32059002004080号
苏公网安备32059002004080号


