
以下内容原文发布于AACR官方博客《Cancer Research Catalyst》, 中文内容仅做参考,请点击文末“阅读原文”,阅览原文内容。
科学方法的基础是对材料、变量和流程的精心控制。实验组之间的任何偏离都可能导致偏倚,从而影响实验结果的有效性。
在实验室层面,这是一条再简单不过的原则,但在临床实践中,事情却很少像这样非黑即白。接受癌症新疗法的患者本身就是异质性的,他们有着不尽相同、种类繁多的合并症和病史,可能导致他们对治疗的应答复杂化。
因此,设立严格的临床试验资格标准有助于确保合并症患者不会因药物的副作用而受到意外伤害,同时也能最大限度地降低潜在有效的药物被认定为无效的几率。然而,这种做法也可能会阻止一些患者接受有助于挽救他们生命的癌症治疗,而且当药物上市时,临床医生也可能会因为缺少数据而无法确定该药在临床试验代表性不足的患者群体中的疗效如何。
那么,对研究人员而言,如何在确保药物得到公平测试的同时,还能让它在高度异质的患者群体中依然安全有效?
最近的几项研究展现了放松入组限制带来的益处,而美国食品和药物管理局(FDA)也紧随其后发布了三份指南草案,用于帮助临床研究人员谨慎设计试验的纳入及排除标准。对现状的彻底改革能否有助于在严谨的科学与包容性的临床实践之间取得理想的平衡?
了解更多内容,请阅读以下原文。
Broadening Clinical Trial Eligibility: A Tricky Balancing Act
The scientific method is rooted in the careful control of materials, variables, and procedures. Any deviations between experimental groups may introduce bias that could compromise the validity of the findings.
It’s a simple enough principle when at the lab bench, but at the bedside, things are rarely so black and white. Patients who receive a new cancer therapy are inherently heterogeneous, harboring a myriad of comorbidities and medical histories that can complicate their response to the treatment.
Accordingly, strict clinical trial eligibility criteria can ensure patients with comorbidities aren’t inadvertently harmed by drugs’ side effects while also minimizing the odds that a potentially effective drug is deemed ineffective. However, this practice may prevent some patients from receiving potentially life-saving cancer treatment, and when the drug reaches the market, clinicians may not have sufficient data to determine how well it works in patient populations that were underrepresented in the trial.
So, how do researchers ensure a drug is being tested fairly but is also safe and effective in a population of patients who are anything but uniform?
On the heels of recent studies hinting at the benefits of loosening the reigns, the U.S. Food and Drug Administration (FDA) has issued three draft guidances intended to help clinical researchers mindfully design the eligibility criteria for their trials. Can an overhaul of the status quo help strike the ideal balance between rigorous science and inclusive clinical practice?
The Role of Clinical Trial Eligibility Criteria
In their draft guidances, the FDA stated that, “The purposes of eligibility criteria for clinical trials are to select the intended patient population and reduce potential risks to trial participants.”

FDA Recommendations for Striking a Balance
In Gelderblom’s study, the most common reasons for protocol exceptions were out-of-range laboratory values and exceptions from required laboratory tests, including biopsies. One of the FDA’s three draft guidances, “Cancer Clinical Trial Eligibility: Laboratory Values,” addressed why laboratory tests are a common source of eligibility restrictions and how they can be potentially reconsidered to allow more patients to enroll.
Studies have shown that eligibility criteria based on laboratory values do not vary much between different clinical trials for cancer therapies, suggesting that they may be carried from trial to trial without careful consideration of which comorbidities may actually pose a risk. Further, reference ranges for “normal” laboratory values may differ based on age, race, and ethnicity, which may enhance disparities in enrollment if these differences are not accounted for.
To solve this, the FDA proposes that trialists provide a clear scientific rationale for each restriction and review them as additional data become available (i.e., between clinical trial phases). The FDA also recommends accounting for disease-relevant changes to organ function. For instance, since liver dysfunction may be more prevalent among patients with liver cancer it should not preclude enrollment.
In “Cancer Clinical Trial Eligibility Criteria: Washout Periods and Concomitant Medications,” the FDA raised similar concerns about the exclusion of patients taking other medications or the mandatory delay between the cessation of one treatment and the start of another. While such restrictions are sometimes important to prevent dangerous drug interactions and may be necessary to accurately test a new therapy’s accuracy, broad exclusion criteria are often employed that may unnecessarily hinder enrollment. As older patients, especially those with cancer, are more likely to take multiple maintenance medications than the general population, they may be more likely to be excluded.
Similar to the guidance for exclusion criteria based on laboratory values, the FDA recommends having a specific rationale for each contraindicated drug class, as well as evidence-based mandatory washout periods that account for the pharmacokinetic and pharmacodynamic properties of the treatment being ceased.
Finally, the FDA addressed eligibility criteria that account for a patient’s “performance status,” or their ability to perform normal daily tasks. Patients with a low performance status are often excluded because they may be more susceptible to side effects, which could more negatively impact their quality of life and skew data related to adverse events.
However, performance status scores are subjective, especially in patients over 65, and may not necessarily indicate additional risk, the FDA explained in “Cancer Clinical Trial Eligibility Criteria: Performance Status.” If a treatment more adversely impacts patients with low performance status, the FDA argued, that data should be known so patients and physicians can be best informed when the drug reaches the market. The FDA recommends including patients with moderate to low performance status whenever possible. If necessary for data analysis, researchers may exclude patients with low performance status from efficacy analyses or stratify patients based on performance scores.
Benefits for Patients and Researchers
The study by Gelderblom and colleagues tested how some of the FDA’s ideas might shape patient outcomes in practice.
Sometimes, patients may be considered for eligibility requirement exemptions, wherein qualified physicians review a patient’s situation and decide whether their enrollment poses a significant risk or may affect the data. Gelderblom and colleagues sought to determine how these exemptions—and, by extension, the broadening of clinical trial eligibility criteria—might affect patient outcomes.
Gelderblom and colleagues assessed survival and response rates for 1,019 patients enrolled in the Drug Rediscovery Protocol (DRUP) trial, a Dutch pan-cancer basket/umbrella trial that matches treatment-refractory patients with off-label targeted therapies based on their tumor’s molecular profile; 82 of these patients received a waiver to enroll despite not meeting all eligibility criteria.
Forty percent of patients who received a waiver experienced a clinical benefit, compared with 33% of patients who met all eligibility criteria; median overall survival was also similar (11 months versus 8 months, respectively). Severe side effects affected 39% of patients granted a protocol waiver and 41% of patients without waivers.
“These findings advocate for a broader and more inclusive design when establishing novel trials, paving the way for a more effective and tailored application of cancer therapies in patients with advanced or refractory disease,” Gelderblom said in a press release about the study.
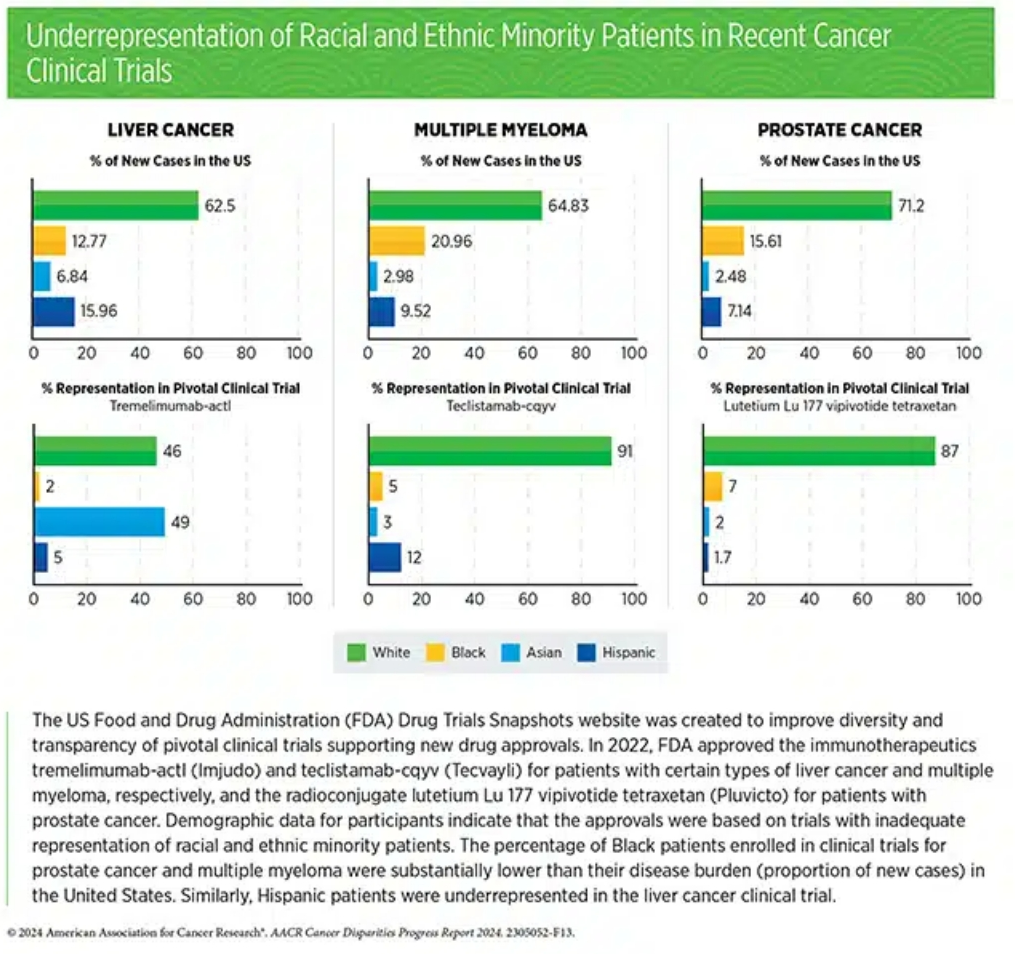
Boosting the number of patients eligible for clinical trials may have additional benefits. In all three draft guidances, the FDA stressed that loosening eligibility criteria may speed up the enrollment process and allow trials to proceed faster. It may also help broaden the diversity of the trial population, a topic tied closely to a related FDA draft guidance issued in 2022 and updated most recently in June 2024.
更多内容,请点击“阅读原文”
排版编辑:肿瘤资讯-Astrid











 苏公网安备32059002004080号
苏公网安备32059002004080号


