
乳腺癌是全球女性最常见的肿瘤,也高居癌症相关死亡原因的首位。对于乳腺癌新靶标和新机制的研究,一直是该领域的探索热点。不过,如何从组学数据提供的大量分子变异确定功能变异依然存在挑战。超过90%人类基因通过可变剪切产生转录本,其中60%的剪切变体,编码出不同的蛋白质异构体。目前已知的癌症十项基本特性都受到异常剪切的调控,而且异常剪切本身已被认为是癌症的基本特征之一,并且是富有临床价值和广阔前景的治疗靶点。核糖核酸(RNA)结合蛋白表达量或活性的变化,能够影响大量癌症相关基因的剪切形式。因此,有必要对乳腺癌中功能性RNA结合蛋白展开系统筛选和深入研究。
2018年6月15日,美国癌症研究学会《癌症研究》正式发表复旦大学附属肿瘤医院、复旦大学上海医学院、中国科学院上海高等研究院、中国科学院上海生命科学研究院的研究报告,首次利用CRISPR/Cas9基因编辑技术对RNA结合蛋白进行系统功能筛选,发现了乳腺癌生存依赖的剪切因子PHF5A,并从可变剪切与表观遗传学角度深入探讨了该分子抑制乳腺癌凋亡的机制,为发掘乳腺癌治疗新靶标提供了理论依据,也为疾病提供了全新的预后标志物。
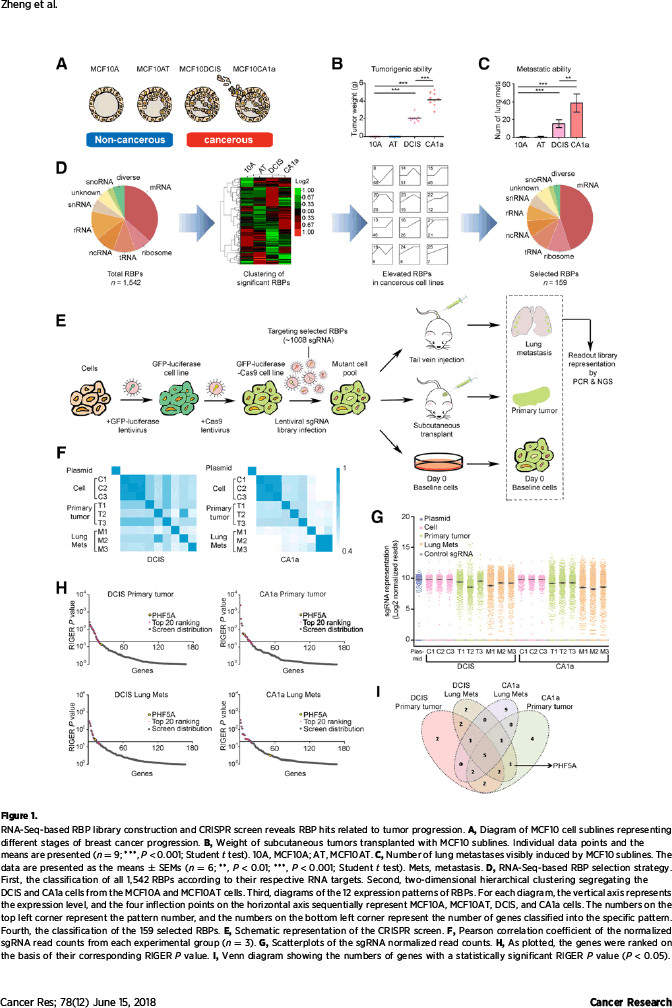
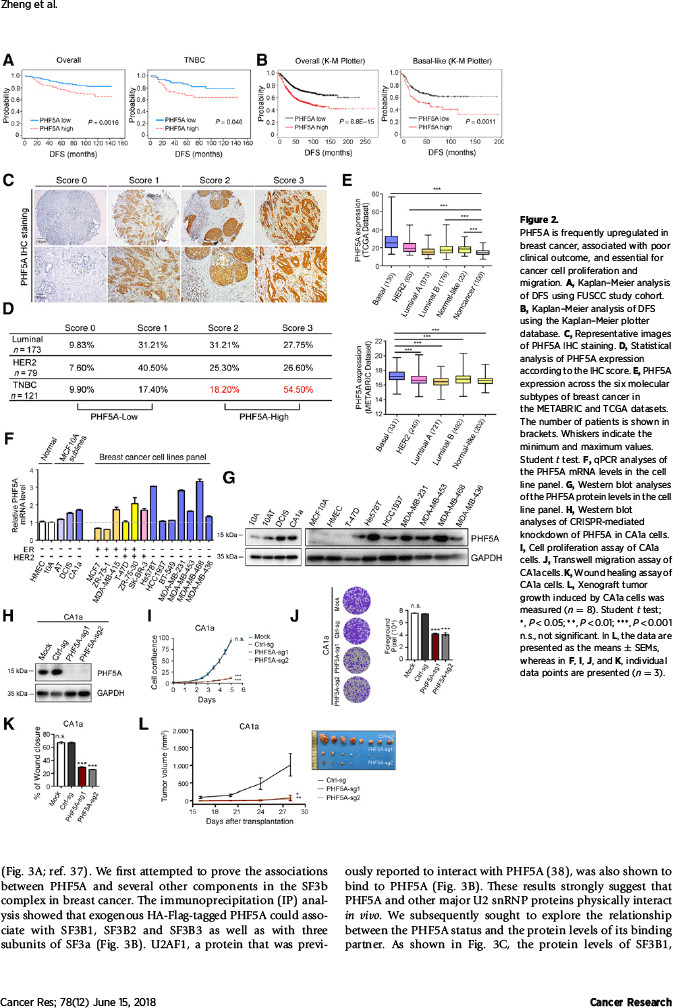
该研究利用CRISPR/Cas9基因编辑技术,自主开展了对RNA结合蛋白的功能筛选,并发现了一批癌细胞生长依赖的重要基因,其中PHF5A在多个实验组中排名高居前列。国际癌症基因组图谱(TCGA)、国际乳腺癌分子分型联盟(METABRIC)、癌症生存曲线分析(Kaplan Meier plotter)以及该中心研究队列表明,乳腺癌组织普遍存在PHF5A高表达,并且其高表达提示预后较差。根据生物学功能,PHF5A缺失可导致癌细胞增殖、迁移和成瘤能力显著受损。
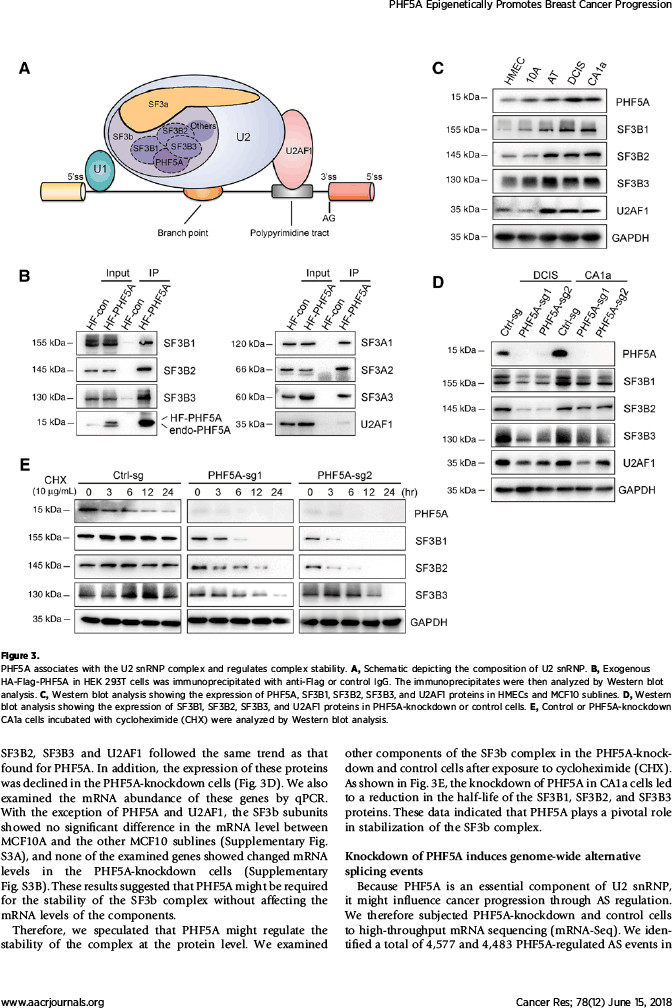
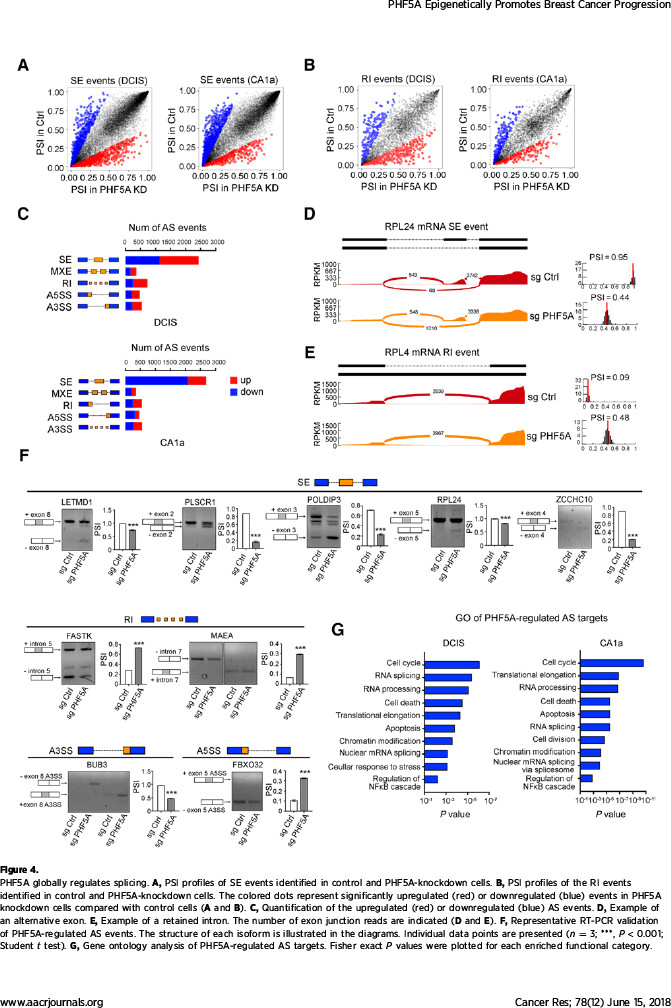
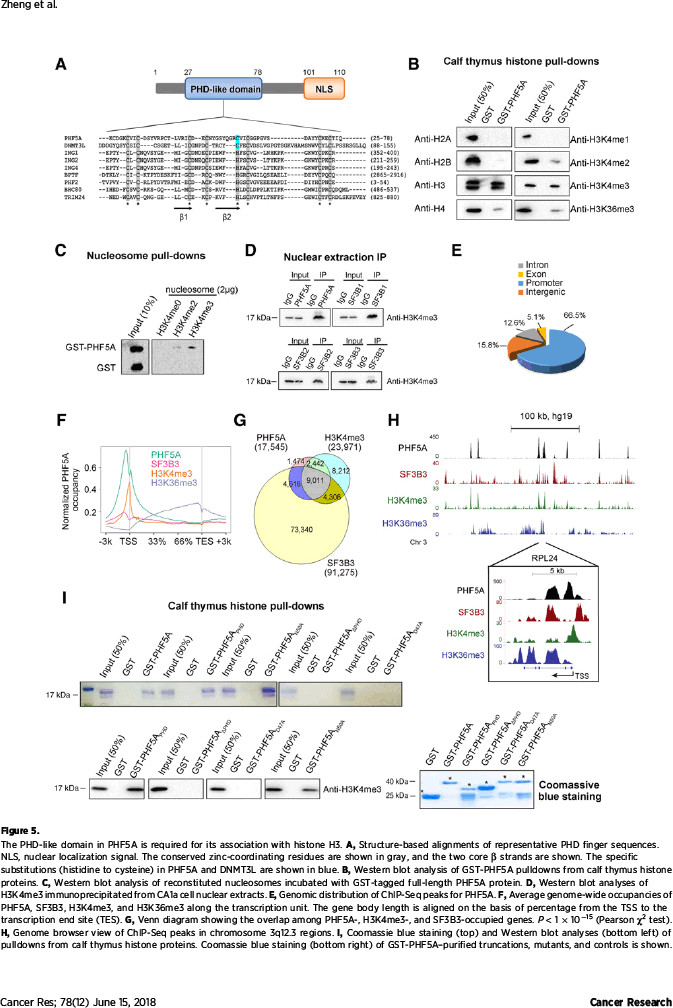
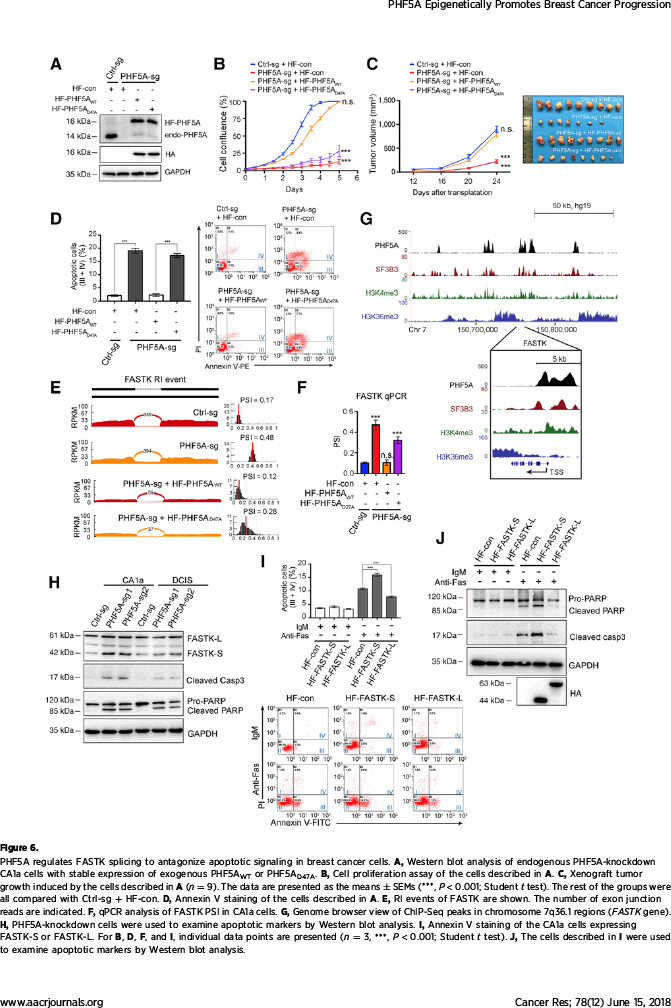
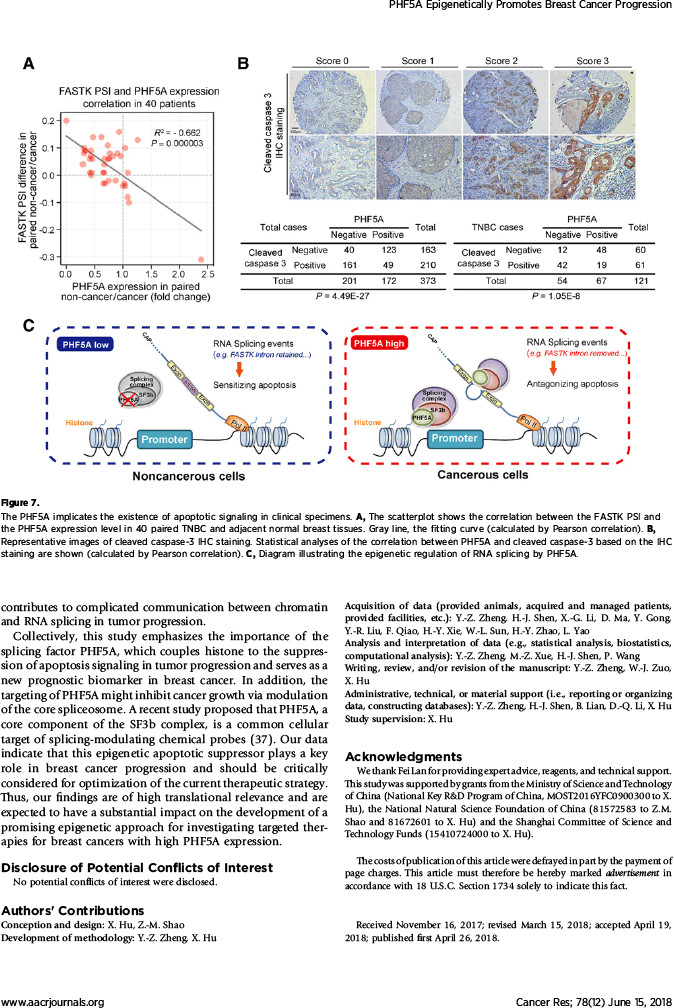
该研究在探索机制过程中,发现了有趣的现象:作为维持U2剪切体复合物稳定性的必需蛋白,PHF5A起着沟通组蛋白修饰信息与转录本可变剪切的桥梁作用。通过自身的PHD指状结构域,PHF5A能够与启动子区域的组蛋白H3相结合,对该部位的基因序列实施可变剪切。该研究发现PHF5A所调控的大量剪切事件在凋亡通路上非常活跃。其中,凋亡相关基因FASTK在PHF5A高表达时主要编码抗凋亡形式的蛋白,而在低表达PHF5A的细胞中编码出促凋亡的截短蛋白。
因此,该研究的重要发现在于筛选获得了功能极为显著的剪切因子,深入揭示了可变剪切与表观遗传学信息的内在联系,为相关领域的研究提供了重要启示,也为乳腺癌新靶标的发掘提供了理论依据。
Cancer Res. 2018 Jun 15;78(12):3190-3206.
PHF5A Epigenetically Inhibits Apoptosis to Promote Breast Cancer Progression.
Zheng YZ, Xue MZ, Shen HJ, Li XG, Ma D, Gong Y, Liu YR, Qiao F, Xie HY, Lian B, Sun WL, Zhao HY, Yao L, Zuo WJ, Li DQ, Wang P, Hu X, Shao ZM.
Fudan University Shanghai Cancer Center, Fudan University, Shanghai, China; Shanghai Medical College, Fudan University, Shanghai, China; Shanghai Advanced Research Institute, Chinese Academy of Sciences, Shanghai, China; Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences, Shanghai, China.
Alternative splicing (AS) and its regulation play critical roles in cancer, yet the dysregulation of AS and its molecular bases in breast cancer development have not yet been elucidated. Using an in vivo CRISPR screen targeting RNA-binding proteins, we identified PHD finger protein 5A (PHF5A) as a key splicing factor involved in tumor progression. PHF5A expression was frequently upregulated in breast cancer and correlated with poor survival, and knockdown of PHF5A significantly suppressed cell proliferation, migration, and tumor formation. PHF5A was required for SF3b spliceosome stability and linked the complex to histones, and the PHF5A-SF3b complex modulated AS changes in apoptotic signaling. In addition, expression of a short truncated FAS-activated serine/threonine kinase (FASTK) protein was increased after PHF5A ablation and facilitated Fas-mediated apoptosis. This PHF5A-modulated FASTK-AS axis was widely present in breast cancer specimens, particularly those of the triple-negative subtype. Taken together, our findings reveal that PHF5A serves as an epigenetic suppressor of apoptosis and thus provides a mechanistic basis for breast cancer progression and may be a valuable therapeutic target. Significance: This study provides an epigenetic mechanistic basis for the aggressive biology of breast cancer and identifies a translatable therapeutic target.
PMID: 29700004
DOI: 10.1158/0008-5472.CAN-17-3514





















 苏公网安备32059002004080号
苏公网安备32059002004080号


